INTRODUCTION
As expected with the discovery of a new disease entity, initial enthusiasm is followed by controversy and a plethora of fundamental questions. In no other recent gastrointestinal disease has this been truer than in eosinophilic esophagitis (EoE). Initial series described young, predominantly men with atopy, characteristic endoscopic features, and response to topical steroids or elimination diets (1,2,3,4). Definitions of EoE have been confounded by heterogeneity in symptom presentations and specificity concerns of eosinophil quantification. Nowhere has this controversy been more apparent than in the distinction of EoE from gastroesophageal reflux disease (GERD). Furthermore, both clinicians and investigators have struggled with agreement on the most pertinent clinical endpoints to define therapeutic response in EoE.
On the other hand, there has been remarkable progress in the understanding of EoE since the time of its recognition two decades ago. Genetic studies have identified specific profiles that support an allergic pathogenesis to the condition (5,6). Prospective and randomized trials have demonstrated the effectiveness and efficacy of topical steroids (7,8,9,10) and of withdrawal of food antigens that trigger the epithelial response (11,12). In addition, endoscopic dilation has provided a generally safe and durable means of ameliorating strictures that complicate the disease (13,14,15,16).
This guideline puts forth recommendations regarding fundamental clinical questions pertaining to the management of EoE (Table 1). Esophageal eosinophilia is emphasized as a conceptual term describing the pathologic finding of increased esophageal epithelial infiltration by eosinophils (eos). This term is highlighted to avoid the etiologic and therefore treatment implications of EoE, and thus places the emphasis on defining the cause of this pathologic finding in individual patients before implementing a specific therapy. This approach is consistent with that of other recent guidelines in the field (17).

Summary and strengths of recommendations and evidence
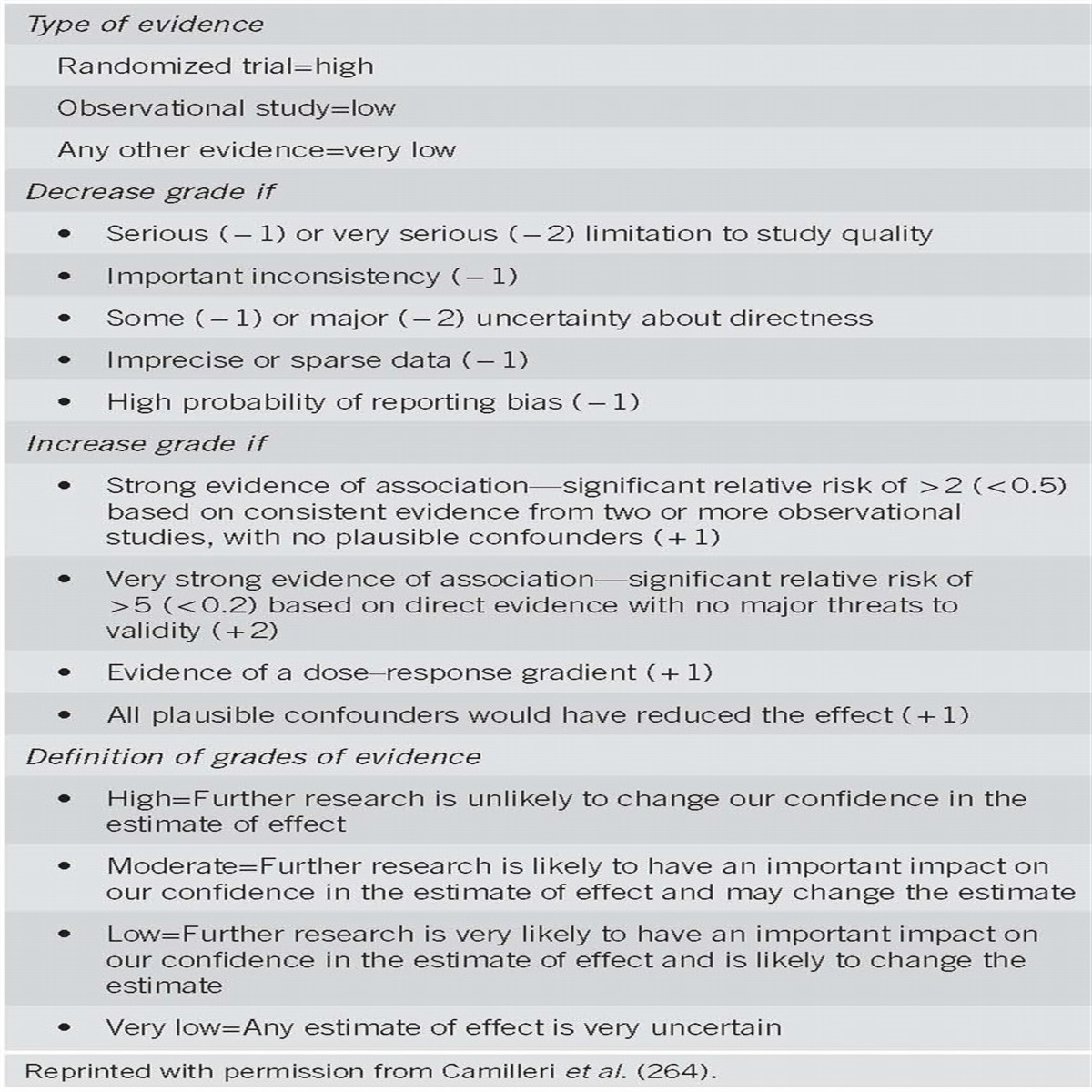
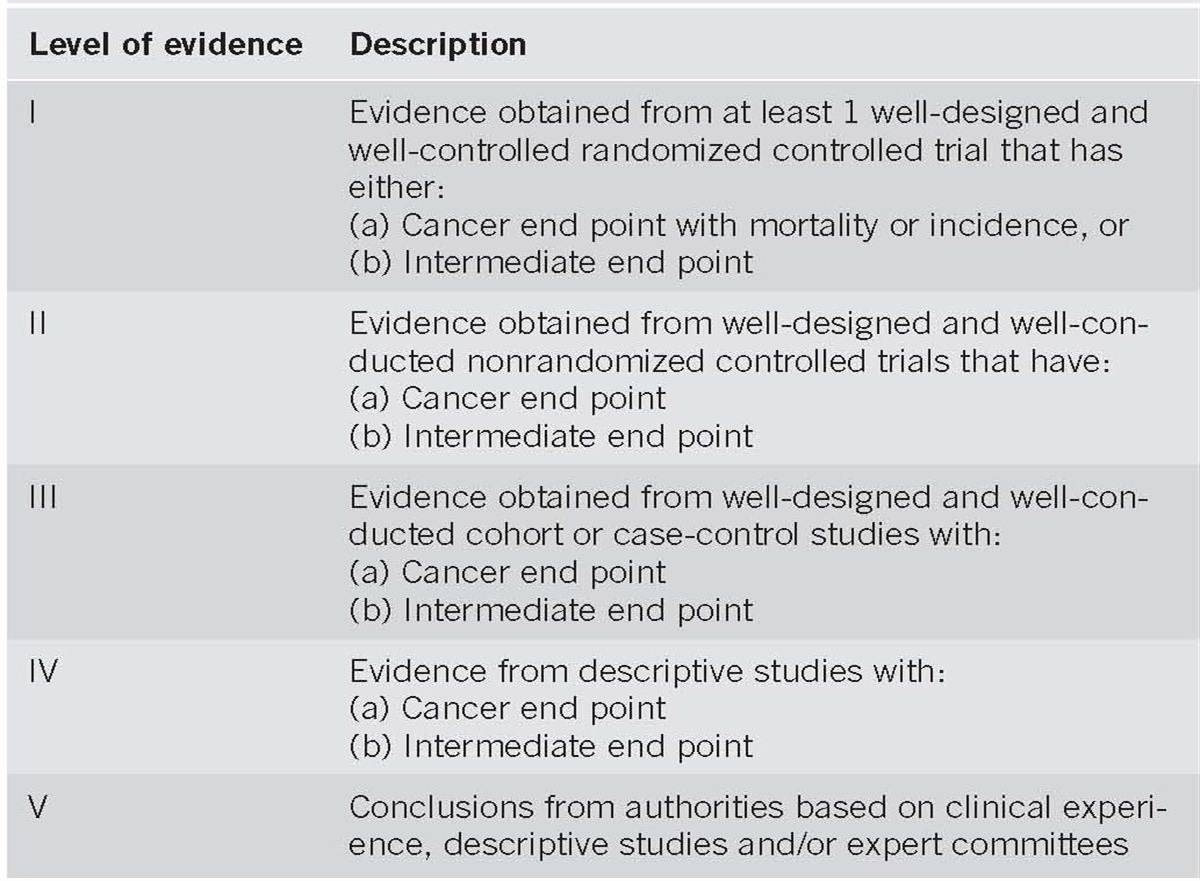
In order to assess the strength of our recommendations and the evidence, the GRADE system was used (18). Recommendations were either strong (desirable effects outweigh undesirable effects) or conditional (trade-offs are less certain), and the quality of evidence was either strong (further research is unlikely to change confidence in the estimate), moderate (further research is likely to change confidence in the estimate), low (further research is very likely to change confidence in the estimate), or very low (the estimate of the effect is very uncertain) (18). In reading this publication, one has to acknowledge that the majority of recommendations are “conditional” rather than “strong”, further emphasizing the paucity of firm data guiding decisions and the likelihood of changing consensus in answer to even some of the most basic questions about this disease.
DIAGNOSIS
Definition and causes of esophageal eosinophilia

No Caption available.
Summary of the evidence.
Our knowledge of the esophageal mucosa is typically limited to information that can be derived from esophageal mucosal punch biopsies. These specimens document the presence of a somewhat bland-stratified squamous epithelium. In health, this surface contains a few lymphocytes but no other leukocytes. During allergic and peptic inflammation, the epithelial surface becomes hyperplastic and accumulates eosinophils (19). Therefore, the current definition of esophageal eosinophilia is the presence of any eosinophils in the esophageal epithelium.
It is important to stress that esophageal eosinophilia is a histological finding that requires interpretation in the clinical context in which it was obtained and that esophageal eosinophilia alone does not define EoE. As an isolated finding, esophageal eosinophilia is most commonly found in three clinical conditions: GERD, EoE, and proton-pump inhibitor-responsive esophageal eosinophilia (PPI-REE), and these cannot be distinguished by the eosinophil count or other associated morphological features (20,21,22). However, a number of other diseases with distinct clinical and histologic features have also been associated with esophageal eosinophilia (Table 2). In approaching these conditions, isolated esophageal eosinophilia must be distinguished from esophageal eosinophilia associated with a more generalized disease such as eosinophilic gastroenteritis or hypereosinophilic syndrome.

Diseases associated with esophageal eosinophilia
Definition of EoE and diagnostic criteria

No Caption available.
Summary of the evidence.
The histologic and phenotypic features of EoE were first described by Attwood (23) and then by Straumann (24). EoE as an allergic disease was first reported in 1995 (25). Originally, EoE was thought to be a rare disease, but over the last 15 years, the prevalence and interest in EoE has greatly increased. The allergic basis of EoE is supported by studies demonstrating that the underlying etiology for EoE is likely an aberrant “antigenic” or “immune” response associated with consistent clinical and histologic abnormalities (26).
Guidelines for the disease were originally written in 2007 and updated in 2011 (17,27). EoE is currently defined as a “chronic, immune/antigen-mediated esophageal disease characterized clinically by symptoms related to esophageal dysfunction and histologically by eosinophil-predominant inflammation” (17). It is also a clinicopathologic disease, meaning that clinical and pathologic information must be considered jointly without either of these parameters interpreted in isolation (17).
Diagnostic criteria: The criteria required for diagnosis of EoE are specified above. Endoscopy with esophageal biopsy is the only reliable diagnostic test for EoE. In the future, translational methods (e.g. RNA microarrays, measurement of specific gene and protein levels via immunochemistry, and/or ELISA) incorporating biologic measures that might be available to refine the definition of EoE, but these are not currently ready for clinical use.
Clinical characteristics: Many studies have described clinical features of EoE, however, none are pathognomonic. The typical EoE patient is an atopic male (male to female ratio 3:1) who presents in childhood or during the third or fourth decade of life (3,28). EoE occurs in most racial and ethnic groups, although many studies have reported predominance in non-Hispanic Whites (29,30). Clinical manifestations of EoE in children are nonspecific and vary by age. Infants and toddlers often present with feeding difficulties, whereas school-aged children are more likely to present with vomiting or pain (31,32). EoE is also commonly associated with other atopic diatheses (food allergy, asthma, eczema, chronic rhinitis, environmental allergies) (27). In adults, solid food dysphagia is the most common presenting symptom (28,33), and food impaction necessitating endoscopic bolus removal occurs in 33–54% of adult EoE patients (34). Other symptoms in adults include chest pain, heartburn, and upper abdominal pain (35).
Physical examinations are useful in children to identify normal growth patterns, and in both children and adults to identify comorbid allergic diseases; however, no features on physical examination are specific in making the diagnosis of EoE.
Endoscopic findings: Endoscopic abnormalities in patients with EoE include fixed esophageal rings (also referred to as a corrugated appearance or trachealization), white exudates or plaques, longitudinal furrows, edema (also referred to as mucosal pallor or decreased vascularity), diffuse esophageal narrowing, and esophageal lacerations induced by passage of the endoscope (a manifestation of mucosal fragility) (17,36,37,38,39). However, because these endoscopic features have been described in other esophageal disorders, none can be considered pathognomonic for EoE.
A meta-analysis of endoscopic findings in EoE from 100 publications encompassing a total of 4,678 patients with EoE and 2,742 controls found that the sensitivity, specificity, and predictive values of endoscopic findings alone are insufficient for diagnosis of EoE (39). In addition, the inter- and intra-observer reliability of detecting these findings is only in the fair range (40), and the endoscopic appearance may be normal in 10–25% of patients with EoE (41,42,43). Therefore, mucosal biopsies of the esophagus should be obtained in all patients in whom EoE is a clinical possibility regardless of the endoscopic appearance. Utilization of a newly validated classification and grading system for endoscopic findings of EoE may improve diagnostic utility (44). This system allows for more uniform characterization of endoscopic findings, facilitates comparisons of severity among clinicians, and provides information regarding fibrostenotic complications of EoE (Table 3). Emerging techniques, such as functional luminal imaging to measure esophageal compliance, may also have clinical utility (45).

Radiologic findings: Strictures, fixed rings, diffuse corrugation, and rarely esophageal intramural diverticulosis have also been described in EoE, but these features are also not specific (17). Esophagrams are not recommended as routine diagnostic tests for EoE. In selected situations, however, they may be useful to characterize anatomic abnormalities such as subtle strictures or small-caliber esophagus that can be difficult to appreciate endoscopically or to provide information on the length and diameter of esophageal strictures for purposes of planning dilation.
Approach to obtaining biopsies: Because inflammatory changes in EoE are frequently patchy and may not be present in all biopsies (46,47), it is recommended that 2–4 biopsies be obtained from at least two different locations in the esophagus, most typically in the distal and proximal halves of the esophagus. It is also reasonable to target esophageal biopsies to the areas with abnormal findings (i.e., rings, plaques, furrows). Several publications have addressed the question of what is the optimal number of mucosal biopsies that should be obtained to maximize the diagnostic yield of EoE (19,46,48,49,50,51,52), and the overall conclusion is that an increasing number of biopsies increases diagnostic yield. When the number of biopsies reaches 6–9, diagnostic sensitivity approaches 100% (46,51,52). Having at least two locations represented in separate pathology jars is helpful for diagnosis not only to assess the extent of inflammation, but also because levels of eosinophilia can vary between the distal and proximal esophagus (7,51,53).
In addition to esophageal biopsies, biopsies of the gastric antrum and duodenum should be obtained once in all children to exclude other potential causes of esophageal eosinophilia. These should also be obtained in adults when there are abnormal endoscopic findings in either the stomach or duodenum or when other gastric or small intestinal symptoms or conditions such as celiac disease or eosinophilic gastroenteritis are clinical possibilities. There are limited data to support routine gastric or duodenal biopsies in adults in the absence of symptoms or endoscopic abnormalities.
Histologic characteristics and diagnostic threshold: While no study has determined an “exact” threshold number of eosinophils that establishes a diagnosis of EoE, there is consensus that finding ≥15 eosinophils in at least one microscopy high-power field on esophageal biopsy specimen after a PPI trial is consistent with the diagnosis of EoE in the proper clinical setting (17). There are some limitations in quantifying eosinophil counts, including lack of standardization of the size of a high-power field (54) and variability in the definition of an intraepithelial eosinophil in hematoxylin-stained tissue sections (47), so communication with a pathologist when there is a question about the results can be helpful. Similar to symptoms and endoscopic findings, elevated esophageal eosinophil counts alone are also not specific for EoE (17,21).
It is important that histologic features besides the absolute eosinophil count, such as eosinophil microabscess formation, superficial layering of eosinophils, extracellular eosinophil granules, basal cell hyperplasia, rete-peg elongation, subepithelial lamina propria fibrosis, and increases in other cell types, such as lymphocytes, be evaluated and noted in pathology reports (47). Although these features are not specific to EoE, they do add information to the overall clinicopathologic assessment of the patient. While preliminary data suggest that the presence of extracellular eosinophil granules (eosinophil peroxidase, major basic protein, and eosinophil-derived neurotoxin) is a useful feature for histological distinction of EoE from GERD (55,56,57,58), special stains have not yet been validated for routine clinical use.
Diagnostic challenges: PPI-responsive esophageal eosinophilia and GERD

No Caption available.
Summary of the evidence.
There are two clinical situations where patients with esophageal eosinophilia respond to PPI therapy. The first is where patients with classic symptoms of reflux and erosive esophagitis or Barrett’s esophagus on endoscopy are found to have esophageal eosinophilia on biopsy (20,59). These patients likely have GERD contributing to esophageal eosinophilia and treatment of their GERD will result in reduction of the eosinophils. The second group of patients present with symptoms suggestive of EoE and may have endoscopic features of EoE, yet their symptoms and esophageal eosinophilia resolve after a PPI course (60). This latter group is now termed PPI-REE (17).
An initial report of PPI-REE by Ngo et al. described three patients with the phenotypic appearance of EoE and high levels of esophageal eosinophils who had complete resolution of esophageal eosinophilia with PPI therapy (20). Since that report, several pediatric and adult studies have described a histologic response to PPI therapy that is consistently in the 30–50% range (60,61,62,63). Stated another way, more than one-third of all patients with esophageal eosinophilia on biopsy will respond to a PPI, and patients in this category should not be diagnosed with EoE.
The reason for this PPI response is incompletely understood and is likely a complex interplay of multiple factors. One possibility is that in GERD, the esophageal epithelium may have damage to the tight junctions due to acid exposure. This results in increased permeability with dilation of intracellular spaces and may allow for allergen penetration, which triggers subsequent recruitment of eosinophils to the esophageal epithelium (64). Another possibility is that there may be a direct anti-inflammatory effect of the PPI on the esophageal epithelium. A recent report has shown that in esophageal cell lines, exposure to omeprazole in cells stimulated with cytokines such as IL-13 and Il-4 can block the secretion of eotaxin-3, which is thought to play an integral role in development of EoE (65).
At present, it is unknown whether patients with PPI-REE represent a GERD variant, an EoE variant, or a separate process entirely. PPI-REE has not been shown to be associated with an antigenic or immunologic cause of esophageal eosinophilia and cannot be labeled as an EoE phenotype at this time. However, long-term followup of these patients is lacking. One small case series highlighting this issue follows four pediatric patients with PPI-REE (63). Despite continued therapy with PPI, patients developed symptoms warranting repeat endoscopy, which ultimately demonstrated recurrent esophageal eosinophilia consistent with EoE. Further longitudinal studies are needed to address this concern and inform future recommendations.
A proposed algorithm of initial treatment and evaluation of esophageal eosinophilia is given in Figure 1, and a PPI trial is central to this. Few data exist to guide specific recommendations on dosage and duration of proton-pump inhibitors as initial therapy for esophageal eosinophilia. Retrospective data support the use of either once or twice daily use, but many of the studies that reported on PPI-REE used twice daily PPI dosing in the 20–40 mg range of several available PPIs (61,62,66). It is recommended that doses should at least be similar to those used to treat GERD-related erosive esophagitis, with a duration of 8 weeks continuing until the time of the follow-up endoscopy and biopsy.

After a patient is found to have PPI-REE, a clinician may choose to continue the evaluation to determine whether GERD is the cause, given that a PPI response may not be specific for reflux (67). Recent guidelines on the evaluation of GERD have been published and can direct this evaluation, and ambulatory pH monitoring may be used in selected patients (68). It is important to note, however, that two studies have found that pH monitoring at baseline reliably whether a patient with esophageal eosinophil responds to a PPI trial (60,69).
TREATMENT
Endpoints of treatment in EoE

No Caption available.
Summary of the evidence.
Complicating the issue of therapeutic endpoints in EoE is that few data specifically examine the degree to which the eosinophil density need be reduced to reverse or prevent ongoing esophageal injury. Accordingly, treatment endpoints chosen in the literature are both variable and arbitrary, with investigators differing in terms of defining the upper limit of eos/hpf to determine a complete response to therapy. For example, histologic endpoints have varied from 0–1 eos/hpf [refs (10,49,70,71,72)] to 0–6 eos/hpf [ref. (9)], a decrease of >90% of eosinophils (8), and have been combined with another histologic parameter such as basal-zone hyperplasia (7). In several studies, the concept of a partial histologic response is also used with variable definitions (9,10,49). While most investigators use a peak eosinophil count, some have reported the mean count (17,19), though these two measures highly correlate with each other(73).
Defining symptom endpoints is also challenging because symptoms of EoE are nonspecific and can be minimized by dietary modifications that can be difficult to quantify. Several randomized clinical trials evaluating budesonide, fluticasone, or prednisone have demonstrated that there is an overall correlation of histologic and symptomatic response to therapy (7,9,10,71). However, there are other studies of EoE treatments where the histologic response does not match with the symptom response (8,74,75,76). Scoring systems for EoE have been developed, but have not been validated for use as outcome measures as of yet (31,77,78). These scoring systems are also mixed as to whether they do or do not correlate with eosinophil counts (31,79). Defining evidence-based treatment endpoints in EoE is an area of active research that will inform future recommendations, and the optimal endpoints of therapy of EoE for the purpose of clinical trials have yet to be defined.
Pharmacologic treatments

No Caption available.
Summary of the evidence.
Topical steroids: Topical corticosteroids have been proven to be an effective therapy for EoE, and are a first-line therapy. The medications, available as multi-dose inhalers or aqueous nebulizer solutions for use in asthma, are swallowed rather than inhaled to coat the esophagus and provide topical medication delivery. Dose ranges are presented in Table 4.

Topical steroid initial dosing for treatment of EoE
In children, randomized trials comparing fluticasone to prednisone (7) and to placebo (71) have demonstrated an approximately 50% complete and 95% partial response using 1–3 months of therapy. Symptomatic and endoscopic improvement was also robust. In the only placebo-controlled trial of fluticasone in adults (8), there was a histologic response in 62% but not a significant improvement in symptoms. For using fluticasone, the patient is directed to puff the inhaler into the mouth during a breath hold, and then to swallow it. After dosing, patients should avoid eating or drinking for 30–60 minutes.
Budesonide has also been proven to be an effective therapy for EoE in randomized trials. In children, aqueous budesonide has been mixed with a sugar substitute (1 mg/2 ml of budesonide with 5 gm of sucralose) to create a slurry termed “oral viscous budesonide” (80,81). A randomized trial of oral viscous budesonide in children showed significant improvement in symptoms, endoscopic findings, and esophageal eosinophilia compared with placebo (9). In adults, a randomized trial of budesonide, which was nebulized and then swallowed showed similar results (10). There have been no studies to date comparing the efficacy of fluticasone to budesonide, but a recent study examined two topical formulations of budesonide (75). In this randomized, open-label trial, the oral viscous form of budesonide was found to be more effective than the nebulized swallowed formulation for decreasing eosinophils counts, likely due to prolonged esophageal dwell time.
Topical steroid therapy is felt to be safe in general. However, candidal esophagitis has been reported in 5–30% of cases, though many times this was incidentally noted on follow-up endoscopy. Oral candidiasis has been reported in only approximately 1% of patients treated with topical steroids regardless of the medication formulation, dose, or whether the mouth was rinsed after medication administration (7,8,9,10,53,71,75,78,80,82,83,84,85). To date, there has been no evidence of adrenal suppression up to 2 months of treatment. Long-term safety data are not yet available for growth rates or bone density.
Systemic steroids: There is only one randomized trial of prednisone for treatment of EoE in children, and the comparator was swallowed, topical fluticasone (7). Although results were similar between the two agents, prednisone led to more complete histologic improvement as well as more adverse events (40% of patients developed side effects such as hyperphagia, weight gain, and/or Cushingoid features). A single cohort study of 39 children with EoE treated with prednisone demonstrated a reduction in eosinophil counts from 34 to 1 eos/hpf after 4 weeks of treatment (53). Because of the potential for side effects, prednisone is typically reserved for times when topical steroids are not effective or patients need a rapid improvement in symptoms.
Other agents: If a patient does not respond to a topical or systemic steroid after dose escalation or a longer course of treatment, a non-pharmacologic treatment of EoE, such as dietary elimination or endoscopic dilation, is recommended. A number of other agents have been studied on a limited basis in EoE but their efficacy is not established, and they are not recommended for use.
There are no controlled trials of leukotriene inhibitors in patients with EoE. In a case series of adults treated with high doses of montelukast, there was symptomatic but not histologic response in 12 patients (86). In a study of 11 adult patients, montelukast was not effective for maintaining a steroid-induced remission (87).
Although mast-cell inhibitors have a theoretical place in the treatment of EoE (88,89,90,91), there are no controlled trials of this form of therapy in EoE. In a case series, cromolyn sodium was used for 4 weeks in 14 children who failed to demonstrate either symptomatic or histologic improvement (53).
The immunomodulators 6-mercaptopurine and azathioprine were used in three adult patients with steroid-dependent esophageal eosinophilia with resulting symptomatic and histologic remission (92). Due to potential side effects and few data, these medications are not recommended for routine use.
Interleukin-5 (IL-5), a key cytokine in eosinophil physiology, is an attractive target for anti-eosinophil therapies (26). Antibodies to IL-5 have been studied, but they are not available for clinical use. Mepolizumab has been used in two trials, one in children and one in adults (93,94). In both, this drug reduced eosinophil counts in most patients, but complete histologic resolution occurred in only a small percentage. In the adult trial, there was no change in symptoms. Reslizumab was used in one trial and demonstrated significant improvement in esophageal eosinophilia in patients with EoE without serious side effects (76). However, the symptomatic response was no different than placebo. Further studies utilizing these medications are warranted with to define their role in EoE.
Omalizumab, an anti-IgE antibody, was not effective in a case series of two subjects (95).
Dietary treatments

No Caption available.
Summary of the evidence.
Strategies for dietary elimination: Three strategies of dietary therapy have evolved. The first is total elimination of all food allergens with elemental or amino–acid-based formula. The second is a targeted elimination diet guided by allergy testing, typically skin prick testing or patch testing. The third is an empiric six-food elimination diet removing the six most common known food groups that are triggers of EoE: soy, egg, milk, wheat, nuts, and seafood. All three approaches have demonstrated symptomatic and histologic resolution in pediatric patients in uncontrolled studies (49,53,96). The duration of the treatment is usually 4–8 weeks, followed by a reintroduction period once remission has been achieved. Because dietary elimination and identification of food allergens is labor, cost, and time intensive, the decision to pursue this strategy should be individualized based on available resources and patient and family preferences.
While an elemental formula has been shown to be the most effective dietary therapy (95–98% resolution of symptoms and histology within 4 weeks) (25,53,97,98), there are some practical limitations to this approach. Elemental formulas are costly, often require the placement of feeding tubes for formula administration, and may impact the quality of a patient’s life. An alternative approach that addresses removal of the most common food allergens without performing allergy tests is the six-food elimination diet. This was developed by Kagalwalla et al. (49) who reported symptomatic and histologic resolution in 74% of a pediatric cohort. Subsequent studies from this group also implied that the most common triggers of EoE upon reintroduction of foods were milk, wheat, egg, and soy (49).
Food elimination has not been as widely studied in adults. Simon et al. attempted a targeted elimination diet based on IgE sensitization to rye and wheat in a small subset of patients without evidence of histologic or symptomatic response (99). Gonsalves et al. conducted a prospective trial of a six-food elimination diet in 50 adult patients for a duration of six weeks (11). Overall, 64% of patients had peak eosinophil counts ≤5 eos/hpf and 70% had peak eosinophil counts ≤10 eos/hpf. Symptoms improved in 94% of patients and endoscopic features improved as well. Systematic reintroduction of food groups identified wheat (60%) and milk (50%) as the most common triggers.
Two additional preliminary adult studies have been presented. Peterson et al. conducted a small trial with elemental formula in 21 adults and found that 48% had <8 eos/hpf after treatment for 4 weeks (100). Lack of adherence with the elemental formula was thought to contribute to the lower response rate when compared to prior pediatric data. Lucendo et al. (101) studied 64 patients who completed a diet eliminating wheat, corn, milk, eggs, nuts/peanuts, legumes, fish/shellfish, rice, and soy. They found 73% of patients had eosinophil counts <5 eos/hpf after dietary elimination. Food reintroduction identified the most common triggers as milk, wheat, and legumes.
The goal of dietary therapy is not only to induce clinical and histologic remission but, importantly, to maintain remission by elimination of a specific dietary trigger(s). For children, maintenance of normal growth and development with a nutritionally replete diet is extremely important. Patients may be offered treatment with dietary therapy as an alternative to chronic pharmacologic therapy if they are motivated to try to find their food trigger(s) and willing to eliminate the food from their diet. Initiation of dietary therapy for EoE often begins with a consultation with a dietitian familiar with food allergies and eosinophilic gastrointestinal disorders. The dietitian should have patients complete a dietary log early in the trial to assess for sources of contamination. Involvement of a dietician is especially important if dietary therapy with elemental formula is to be pursued in order to make sure that adequate caloric intake is being met and to assess for any potential electrolyte or micronutrient imbalance.
After allergens are removed from the diet, food reintroduction is required to determine the specific triggers of EoE. A limited amount of data as well as clinical experience provide practical guidance to the reintroduction process. The decision of which food(s) to add is often made as a collaborative effort between the patient, family, and physicians. Some studies recommend that one food or food group is introduced every 4–6 weeks with observation of clinical symptoms and a subsequent endoscopy if no change in symptoms occur (70,102,103), while others have reintroduced combinations of foods (98). In the Gonsalves study (11), one food was added back every 2 weeks and endoscopy was performed after two foods. A food trigger is identified based on the recurrence of symptoms and esophageal eosinophilia (e.g., >15 eos/hpf) after reintroduction of a specific food group. Because patients typically have more than one food trigger, the process is continued until all foods have been added back or an acceptable diet is reached. Patients who are initially treated with an elemental formula undergo a substantially longer reintroduction process. Once food triggers are identified, patients are advised to eliminate these agents from their diet completely. Practical factors to consider when using this approach include the cost of and risk associated with repeated endoscopy. Long-term maintenance studies of the efficacy of dietary therapy are warranted, as are prospective studies to determine the optimal approach to food reintroduction.
Role of the allergist: The use of allergy testing to identify foods that cause EoE is controversial. There are three major types of allergy tests: immunoassays for serum food-specific IgE; skin prick testing; and atopy patch testing. Currently, the best approach for employing these modalities in patients with EoE has not been determined and no single modality has been universally adopted. Data supporting an allergy testing and targeted elimination approach exist in children at some centers (103) but not at others (98), and skin prick testing was only 13% predictive in identifying food allergens in adults (11).
Reasons for this include variability in subject selection, antigens examined, and technical differences in skin-testing techniques and readouts. Future multicenter studies focused on standardizing and validating these tests are needed.
There are important roles for allergists in treating patients with EoE including the identification, treatment, and evaluation of allergy testing in EoE patients, as well as the assessment of comorbid allergic diseases such as atopic dermatitis, asthma, and allergic rhinitis/conjunctivitis (17). Allergists can also identify other food allergies, such as those that are IgE mediated, and provide prophylactic treatments and instructions for anaphylaxis. As these are outside of the areas of expertise of most gastroenterologists, a close collaboration can be helpful. At some centers, not only do allergists take primary responsibility for the management and coordination of care of EoE patients, but they perform important clinical and basic research on EoE. Finally, dieticians can also play an important role. Their knowledge of overlap and cross contamination of specific food items and expertise in coping with a restricted diet from a compliance, social, economic, and practical point of view are often very helpful when using dietary therapy in patients with EoE.
Endoscopic treatment

No Caption available.
Summary of the evidence.
The fibrostenotic complications of EoE include focal esophageal strictures and narrow-caliber esophagus (13,17,104). Esophageal dilation is an effective treatment for such changes, and was one of the first therapies used for adult patients with EoE (17). In several large series, esophageal dilation relieved dysphagia in majority of patients (14,15,105,106). Moreover, the mean duration of response to dilation was more than a year (105). There was also a very high degree of patient acceptance for primary therapy with esophageal dilation in a post-dilation survey, with all patients willing to undergo repeated dilation as needed (105). In general, it is preferable to reserve dilation until after the effects of medical or dietary therapy can be assessed (17). However, on an initial endoscopy, if a critical stricture is encountered or food impaction has occurred, then dilation can be performed prior to other therapies. The role of dilation as a primary monotherapy of EoE is still controversial and should be individualized until more data are available.
Dilation technique: Few data exist to support one specific method of esophageal dilation over another. While a through-the-scope hydrostatic balloon dilator offers the theoretical advantage of avoidance of shear stress injury that is delivered by bougie dilation, there is no convincing evidence that balloon dilation offers greater safety in EoE. Hydrostatic balloon dilation does allow for inspection of the underlying esophageal mucosa between serial dilations without the need to reintroduce the endoscope. Bougie dilation using a wire-guided system offers the ability to dilate multiple strictures and long strictures such as in a diffusely narrowed esophagus in EoE.
We advocate a conservative approach to dilation, with relatively small increases in diameter over multiple sessions, primarily related to the known esophageal mucosal fragility in EoE (37). After a mucosal rent, disruption, or laceration is noted, no further dilation is performed in that session. This approach encourages mucosal assessment after each incremental dilator size, regardless of whether balloons or bougies are used. Choice of the initial dilator size is important. If the regular adult upper endoscopy passes without resistance (typical diameters range from 9–10 mm), then an initial dilator size just above this may be selected. Resistance to the passage of the bougie may offer an estimate of the degree of luminal compromise that can be difficult to assess endoscopically. An eventual goal of 15–18 mm for esophageal diameter after dilation in EoE has been reported to provide lasting relief of dysphagia, but this target is based on retrospective data and expert opinion, and typically requires multiple sessions to achieve (105,107).
Risks of dilation: Early reports of severe complications related to esophageal dilation in EoE generated trepidation among gastroenterologists (108). Further compounding this concern were early reports of esophageal tears and perforations during endoscopic treatment of food impactions and diagnostic endoscopies, suggesting a marked esophageal fragility in EoE (37,109). An important factor that may have influenced these high complication rates was a lack of disease awareness and perhaps, overly aggressive dilation technique. Specifically, many of the initial reports of esophageal perforation occurred in patients in whom EoE was not initially recognized and prior to publications describing the potential risks of esophageal dilation (16,107,110).
Three recent retrospective studies from adult centers reported complication rates that were considerably lower than that described in initial reports (14,15,105). In two recent meta-analyses of esophageal dilation in EoE, the perforation rate for dilation was 0.3% (107,110), similar to that quoted for esophageal dilation for other benign esophageal diseases. It is important to note, however, that this rate was achieved at academic centers experienced in performing esophageal dilation in patients with EoE. Interestingly, the most common risk of dilation in EoE is post-procedural chest pain, reported in almost three quarters of patients when asked about this symptom prospectively (105). Major bleeding defined by need for endoscopic hemostasis or blood product is rare (only 1 patient reported to date). Nevertheless, the greater safety reported in more recent series may reflect the adoption of a more conservative approach by gastroenterologists who are aware of the potential hazards of dilation in EoE.
OUTCOMES
Natural history of EoE

No Caption available.
Summary of the evidence.
While natural history data on EoE patients are limited, information can be gleaned from multiple sources, including a prospective cohort (13), placebo arms of randomized clinical treatment trials (8,9,10,71,76,85,93), and multiple retrospective case series (12,53,77,84,111,112,113). It is clear that EoE is a chronic disease (17,114), and in many cases, particularly in adults, there can be a decade or more of symptoms prior to diagnosis (105). In the placebo-controlled RCTs conducted to date, endoscopic findings and esophageal eosinophilia typically persist in the placebo arms (8,9,10,71,76,85,93). To date, no case of EoE has been noted to progress to hypereosinophilic syndrome or malignancy (12,77,93,111,112,114).
Cross-sectional symptom and endoscopy data suggest that some patients with EoE may progress from an inflammatory to a fibrotic process, and this could explain differences in presentation and findings between children and adults with this condition (39,77,84,115). These clinical observations are consistent with mechanistic studies showing fibrosis and remodeling in the esophagus in some children (116,117).
A number of studies show that EoE recurs nearly universally after treatment is withdrawn, although the time course may vary for individual patients. From an early report of recurrent symptoms and esophageal eosinophilia after systemic corticosteroids were discontinued (118), recurrence of symptoms in EoE has been a common theme (17) and has also been seen with swallowed topical corticosteroids (119,120), leukotriene antagonists (86), and 6-mercaptopurine (92). The sum of prospective and retrospective dietary elimination data also suggest that if allergens are reintroduced, or elimination and elemental diets are discontinued, symptoms, endoscopic findings, and esophageal eosinophilia recur (11,49,53,70,97,113,121).
Maintenance therapy

No Caption available.
Summary of the evidence.
Because EoE is a chronic disease and symptom recurrence is common after discontinuing treatment, it is reasonable to counsel patients about the potential indications and treatment options for maintenance therapy. The clinician must take into consideration the nature of a patient’s symptoms, their severity, prior and potential complications of EoE, the impact of the selected treatment on the patient’s and family’s quality of life, and individual or family resources to support treatment.
Potential indications for maintenance therapy are: narrow-caliber esophagus; prior esophageal stricture requiring repeated dilations; prior emergent endoscopy performed for esophageal food bolus impaction; prior esophageal perforation; prior Boerhaave’s syndrome; severe or ongoing symptoms; and patient preference.
For pharmacologic therapy, there has only been one maintenance study conducted (120). In this trial, 28 adults with EoE who responded to treatment with 2 weeks of nebulized/swallowed budesonide therapy were randomized to receive either a maintenance dose of budesonide (0.25 mg twice daily) or placebo and were treated for 50 weeks. Those who were maintained on budesonide had a modest reduction in eosinophilia and fewer recurrent symptoms when compared to controls, but there was still substantial recurrence at this low dose. In addition, no patients were diagnosed with superimposed viral or fungal infections, and none had evidence of mucosal atrophy. A possible starting dose for maintenance therapy in adults would be 1 mg/day of budesonide or 880 mcg/day of fluticasone, with lower doses in children, and with doses titrated to provide the best clinical response at the lowest achievable dose.
Long-term dietary elimination therapy is a common maintenance strategy, particularly in children with EoE. While there have not been clinical trials of this strategy, large case series with good follow-up intervals support its use in children (12,53), and now in adults as well (11).
Intermittent esophageal dilation, typically in an “on-demand” fashion for recurrent symptoms of dysphagia, is also a possible maintenance strategy. If used alone, this does not have an impact on the underlying esophageal eosinophilia (105), but this strategy can be effective for treating symptoms of dysphagia (105,106).
CONCLUSIONS
Our understanding of esophageal eosinophilia and EoE have substantially advanced over the past two decades. EoE is now a commonly encountered cause of upper gastrointestinal symptoms and morbidity in both children and adults. This guideline addressed the diagnosis and management of these entities, while emphasizing that the isolated finding of EoE does not equate to EoE. From a diagnostic standpoint, the finding of EoE should trigger an evaluation for its cause. Though there is a broad differential diagnosis, the three most commonly encountered gastrointestinal conditions are GERD, PPI-REE, and EoE. A PPI trial is important for distinguishing EoE from PPI-REE, and is now required prior to the formal diagnosis of EoE. Once diagnosed, first-line options for treatment of EoE include swallowed, topical corticosteroids or dietary elimination. The choice of therapy will depend on patient preference, provider experience, and local expertise. Endoscopic dilation is an effective treatment for strictures or narrow-caliber esophagus, particularly when symptoms of dysphagia are refractory to medical or dietary therapy. Because EoE is chronic and symptoms recur when treatments are discontinued, maintenance therapy should be considered in all patients, but particularly in those with fibrostenotic complications or severe symptoms. Because the field is rapidly evolving, we expect that in the coming years there will be shifts in guidelines and recommendations, as new data emerge concerning noninvasive diagnostic strategies with biomarkers or genetic analysis, novel treatment modalities, non-endoscopic methods to monitor treatment response, and long-term outcomes.
CONFLICT OF INTEREST
Guarantor of the article: David A. Katzka, MD, FACG.
Specific author contributions: Planning of the guidelines: Evan S. Dellon, Nirmala Gonsalves, Ikuo Hirano, Glenn T. Furuta, Christopher A. Liacouras, and David A. Katzka; researching of the guidelines: Evan S. Dellon, Nirmala Gonsalves, Ikuo Hirano, Glenn T. Furuta, Christopher A. Liacouras, and David A. Katzka; writing of the guidelines: Evan S. Dellon, Nirmala Gonsalves, Ikuo Hirano, Glenn T. Furuta, Christopher A. Liacouras, and David A. Katzka; editing of the guidelines: Evan S. Dellon, Nirmala Gonsalves, Ikuo Hirano, Glenn T. Furuta, Christopher A. Liacouras, and David A. Katzka.
Financial support: Evan S. Dellon has received grant/research funding from AstraZeneca, Meritage Pharma, ACG, AGA, NIH, CURED Foundation, and educational support from Pentax. Glenn T. Furuta has received grant/research support from Meritage Pharma, Nutricia and the North American Society of Pediatric Gastroenterology, Hepatology and Nutrition. Ikuo Hirano has received grant/research funding from NIH and CURED Foundation. Nirmala Gonsalves has received grant/research funding from CURED Foundation.
Potential competing interests: David A. Katzka, Nirmala Gonsalves declare no conflict of interest. Dellon is a consultant to Oncoscope. Christopher A. Liacouras is a speaker for Nutricia, and is on the American Partnership for Eosinophilic Disorders. Ikuo Hirano is on the advisory board of Meritage and Aptalis.
REFERENCES